March 2026
Welcome to our first newsletter of spring!
In case you missed it, we are actively recruiting for up to three new trustees for our board. If you would like to be actively involved in setting the direction of First Steps and you have experience in: organisation/business management and growth; charity governance; accounting and financial management in the charitable sector; or fundraising, including from trusts and foundations, please do consider applying. You can find out more on our vacancies page.
Please see the contents of our March newsletter below.
NEWS
Infant milk news
BFLG-UK news
Forthcoming
Imperial Ultra-Processed Food Policy Forum, hybrid, March 24th
An update of First Steps’ Eating Well Healthy Start and Best Start Foods, a practical guide
The iHV Evidence Based Practice conference, Bournemouth, May 6th
HR news
News
New report: SACN report on Nutrition and Maternal Weight Outcomes
In February 2026, the Scientific Advisory Committee on Nutrition (SACN) published its first report on nutrition and maternal weight outcomes, reviewing the scientific basis of current UK dietary recommendations for women before pregnancy, during pregnancy and up to 24 months after delivery. This is the first of two publications from SACN on nutrition and maternal health, with a future position statement to follow on wider UK dietary recommendations for women of childbearing age. We at First Steps Nutrition Trust welcome this report and the renewed focus it brings to the importance of nutrition in pregnancy and early life, particularly for those most at risk of poor dietary outcomes.
Report overview
Around half of pregnant women in England are living with overweight or obesity (50.3%) at their first antenatal appointment, and overweight and obesity is increasing among women of childbearing age more broadly. National surveys show that between a third and two-thirds of women aged 16 to 54 years in England were living with overweight or obesity in 2022, with prevalence highest in the most deprived areas. At the same time, around 1 in 10 non-pregnant young women aged 16 to 24 were living with underweight, highlighting that poor nutritional status affects women across the weight spectrum.
The report is clear that the dietary patterns of most women of childbearing age fall short of UK recommendations. Survey data indicates that women, like the UK population overall, are not meeting recommendations for vegetables and fruit, fibre, oily fish, sugar, salt or saturated fat, and that energy intakes are likely to exceed requirements. There is currently no national dietary data set on pregnant or breastfeeding women in the UK, which SACN identifies as a significant gap.
The evidence reviewed by SACN found that improving dietary quality during and after pregnancy has modest but meaningful benefits for gestational weight gain (GWG) and postpartum weight. Lower glycaemic load dietary patterns, Mediterranean-style diets, and healthier eating patterns more broadly were associated with lower GWG. Dietary supplements, however, were not found to support healthier weight outcomes for most women. SACN also reiterates (in line with the 2025 NICE Maternal and Child Nutrition guidelines) that intentional weight loss during pregnancy is not recommended because of potential adverse effects on the baby.
Key Findings and Recommendations
Weight before pregnancy matters most: SACN confirms what we already knew; that a woman's weight at the start of pregnancy has a greater influence on health outcomes for her and her baby than how much weight she gains during pregnancy. The report emphasises the importance of supporting women to reach a healthy weight before pregnancy, and in between pregnancies.
No change to energy advice during pregnancy and breastfeeding: Women should consume an extra 200 kcal per day in the last three months of pregnancy, and around 330 kcal per day during the first six months of exclusive breastfeeding. SACN notes that women living with overweight or obesity may already be meeting their energy needs without eating more.
Breastfeeding remains a priority: Exclusive breastfeeding for the first six months is linked to lower maternal BMI in the longer term. SACN recommends that strategies to promote and support breastfeeding are strengthened, in line with the wider evidence on its benefits for mothers and babies.
Focus on diet quality, not supplements: SACN recommends that women follow existing UK dietary guidance and eat a healthy, balanced diet. The evidence does not support the use of dietary supplements to manage weight gain during pregnancy for most women.
Some groups need particular attention: The report highlights the need to focus on adolescent girls, older mothers, women from racially and ethnically diverse backgrounds, and those experiencing multiple disadvantage. There is a particular lack of evidence on adolescent girls, who are more likely to be living with underweight and whose energy needs in pregnancy are not well understood.
Better data is needed: SACN calls on government to collect nationally representative data on the nutrient intakes of pregnant and breastfeeding women, and to continue measuring and reporting maternal weight at the start of pregnancy. This is a significant gap that makes it harder to understand and respond to the needs of this group.
Looking ahead
This report is a timely and important contribution to the evidence base on maternal nutrition and weight. Its emphasis on improving diet quality before and during pregnancy, and on reaching the most marginalised groups, reflects a clear understanding that individual behaviour change alone is not enough.
The SACN report sits well alongside the Royal College of Physicians' recently published view on maternal health and obesity, which raises many of the same concerns. Both reports highlight the stark inequalities in maternal health outcomes and call for better support for women from the most deprived communities and those from racially and ethnically diverse backgrounds. Both also emphasise the importance of breastfeeding support. The RCP report adds to this by calling for the Healthy Start scheme to be expanded and for payments to keep pace with food price inflation, recognising that financial barriers must also be addressed. Together, these two reports make a strong case for sustained investment in maternal nutrition and for policies that tackle the wider factors shaping women’s diets.
Further resources
For practical advice on meeting nutritional needs during pregnancy and while breastfeeding, see our Eating Well resources, which can be downloaded for FREE or with a donation:
New DHSC guidance: The Healthy Child Programme
On February 6th, the Department for Health and Social Care published the Healthy Child Programme national framework for improving the health and wellbeing of babies and children and young people from birth and aged up to 19-25 years old in England. It sets out evidence-based approaches to prevention, early intervention and family support – supporting the government’s ambition of raising the healthiest ever generation of children. It comprises delivery guidance for public health nursing teams for and their provider organisations (divided in to part 1, principles, part 2 for health visiting (0-5s), and part 3 for school nursing (5-19s)), commissioning guidance and a high-impact area framework (again broken down in to sections, one of which is for health visiting and 0-5s) which translates the principles in to actionable priorities.
In relation to early years nutrition, the health visiting section of the high impact area framework contains the detail about what health visitors are meant to do to support women antenatally and thereafter with breastfeeding, infant feeding and healthy weight and nutrition. It refers to relevant SACN and NICE guidelines, the Unicef UK Baby Friendly Initiative, the International Code of Marketing of Breastmilk Substitutes and also our resources at First Steps Nutrition Trust for supporting eating well from pre-conception up to the age of 5 years.
The guidance does not introduce any new statutory duties; it is about providing long overdue clarity on what should be delivered and how to improve the quality and consistency of services provided under the health child programme across England in line with pre-existing evidence-based recommendations and guidance.
Read more in the iHV blog here.
New paper: Navigating Infant Feeding in the Context of Household Food Insecurity: A Qualitative Study of New Zealand Mothers
This qualitative study by Ioanna Katiforis and colleagues was published in the March issue of the Journal of the Academy of Nutrition and Dietetics, and explores how mothers in New Zealand navigate infant feeding while living with household food insecurity. The paper focuses on the complementary feeding period, when infants are introduced to solid foods from around six months of age. This is an area that has received relatively little research attention compared with breastfeeding and is highly relevant to the UK context, where levels of food insecurity among households with young children are comparable to those in New Zealand. The pressures described by mothers, including rising food prices, high housing costs, and reliance on a mix of statutory and charitable support, also closely mirror those experienced by families with infants in the UK.
Study overview
The study involved in-depth, semi-structured interviews with 15 mothers living in Dunedin, New Zealand, who had been identified as experiencing moderate or severe household food insecurity when their infants were aged 7–10 months. Interviews explored food purchasing practices, infant feeding during the complementary feeding period, and experiences of seeking support. Data were analysed using reflexive thematic analysis.
Three main themes were identified:
1. Food purchasing strategies were used to stretch money
Mothers described using a wide range of strategies to manage limited food budgets. These included shopping around for the lowest prices, prioritising foods on promotion, buying supermarket own-brand products, and relying on frozen or canned fruit and vegetables when fresh produce was unaffordable. Food purchasing decisions were made alongside other unavoidable household costs such as rent, utilities and transport, requiring constant financial trade-offs.
“Food, money, everything like that sort of stuff was always a constant, like, “Do I have enough?” Like, “Okay I’ve done my groceries on the payday, but am I gonna make it through to the next payday?” I hated living week to week, I hated it. I hated it.” – Mother of 4, single-adult household
2. The infant’s nutrition was prioritised
Across interviews, mothers consistently reported prioritising their infants’ nutrition above their own. Infants were given access to the most nutritious foods available in the household, while mothers reduced portion sizes, relied on cheaper filler foods, or skipped meals altogether. Feeding practices generally aligned with complementary feeding guidance, with strong emphasis on fruits, vegetables and home-prepared foods where time and resources allowed.
“Like she was always fed first and had the most nutritional things for her meals and her snacks... she’s always come first kind of thing, and I’d never jeopardize [that] just for me to get a meal or whatever.” – Mother of 1, single-adult household
A related subtheme concerned breastfeeding. Breastmilk was widely viewed by mothers as an important nutritional safeguard for their infants. However, the study highlights that breastfeeding is not cost-free. Mothers described the physical exhaustion, mental load and increased nutritional demands associated with breastfeeding while food insecure, and the mother’s perceived challenges of sustaining breastfeeding when their own diets were inadequate.
“I was trying to feed myself so that I could feed her… I absolutely did not have enough to eat.” – Mother of 1, two-adult household
The authors also note that for mothers who cannot or choose not to breastfeed, the lack of financial support to purchase infant formula may place additional strain on already limited household budgets.
3. Support was appreciated, but seeking food or money was often disempowering
Mothers accessed support from a range of sources, including family members, friends, food banks and government food grants. While this support was often essential in preventing food shortages, seeking help was commonly associated with feelings of shame, loss of independence and being judged, particularly in interactions with welfare services. Some mothers delayed or avoided accessing support despite being eligible.
“You come away feeling really bad about yourself… like, ‘If you’re poor, that’s your fault…’” – Mother of 1, two-adult household
Author conclusions and implications for UK Policy
The authors conclude that reliance on charitable food assistance and one-off food grants is insufficient to support families with infants, and that longer-term, more acceptable forms of support are needed. Several implications are particularly relevant to UK policy and to the work of First Steps.
Nutrition support in infancy: The findings reinforce the importance of nationally funded schemes that support the nutrition of pregnant women, infants and young children. In the UK, the Healthy Start scheme provides an important foundation but remains limited in value given food price inflation, and in reach, particularly for families navigating complementary feeding.
Income adequacy for families with infants: Food insecurity during infancy cannot be addressed without tackling income adequacy. The pressures described in the study reflect the wider cost-of-living crisis, with rising housing, energy and transport costs placing sustained strain on household budgets. These pressures closely mirror those faced by low-income families in the UK who rely on Universal Credit, statutory maternity pay and low wages.
Integration of food support into maternal and child health services: Embedding food and nutrition support within trusted services such as health visiting and Family Hubs may improve access, reduce stigma and better meet infant-specific nutritional needs.
Maternal nutrition: The study highlights the extent to which mothers compromise their own diets to protect their infants’ nutrition. Policies and programmes should more explicitly recognise and support maternal nutritional needs, particularly for breastfeeding women (see our news piece above on SACN’s new report on Nutrition and Maternal Weight Outcomes). In the UK, this includes supporting pregnant and breastfeeding mothers on low incomes to access schemes such as Healthy Start, which can help with the cost of fruit, vegetables and milk, as well as free Healthy Start vitamins providing folic acid, vitamin C and vitamin D.
This study provides valuable insight into mothers’ lived experience of feeding their infants while being food-insecure. Mothers demonstrated high levels of nutritional awareness, skill and commitment, but did so at considerable personal cost. The findings underline the importance of moving beyond emergency food provision towards coherent policies that support both infant and maternal nutrition during the early years.
Further resources
For practical guidance on supporting families with limited resources, see our guide “Eating well for pregnant and breastfeeding women, babies and young children living in temporary accommodation”. Our “Healthy Start and Best Start Foods: A Practical Guide” may also be useful for families eligible for these schemes. Both resources are available on our website for FREE or a donation here.
New paper: Feeding Profit: how the food and drinks industry (FDI) is one of the key drivers of the global epidemic of childhood overweight and obesity
Unicef published ‘Feeding Profit: how food environments are failing children’ in September last year (see our October newsletter). This commissioned view point in BMJ Paediatrics Open, published in January, focuses on the role of the food and drinks industry (FDI) in driving the global epidemic of overweight and obesity.
The authors outline the contributions of the FDI’s marketing practices, financial models and tactics to block market regulation, to wider commercial determinants of child health. The role of the obesogenic environment on children in their early years in shaping later risk of non-communicable diseases is outlined, and the multitude of ways the FDI violates children’s rights is described. The authors highlight key actions Governments can take to transform children’s food environments for the better, including implementing the International Code of the Marketing of Breastmilk Substitutes and enacting legal measure to limit the availability, marketing and purchase of unhealthy foods and drinks and concomitant measures to enable improved availability and affordability of locally produced nutritious foods for children. Of most relevance to readers of this newsletter, they also outline how other stakeholders including civil society groups, academics and child health professionals can exert positive pressure to improve the obesogenic environment, as shown in box 2.
Recommendations include: ending conflicts of interest and sponsorship of professional society meetings and journals by companies promoting unhealthy products for infants and young children; and focusing on the role of the FDI and their marketing techniques in policy documents and clinical guidance, not just on individual behaviour change interventions.
The authors conclude: “The Unicef report is a powerful advocacy tool that confronts the key drivers of child and adolescent overweight/obesity and challenges governments and other stakeholders to reverse the obesogenic environment driven by profit”.
Read the paper here.
Infant Milk News
Updated: Infant milk costs and trends reports
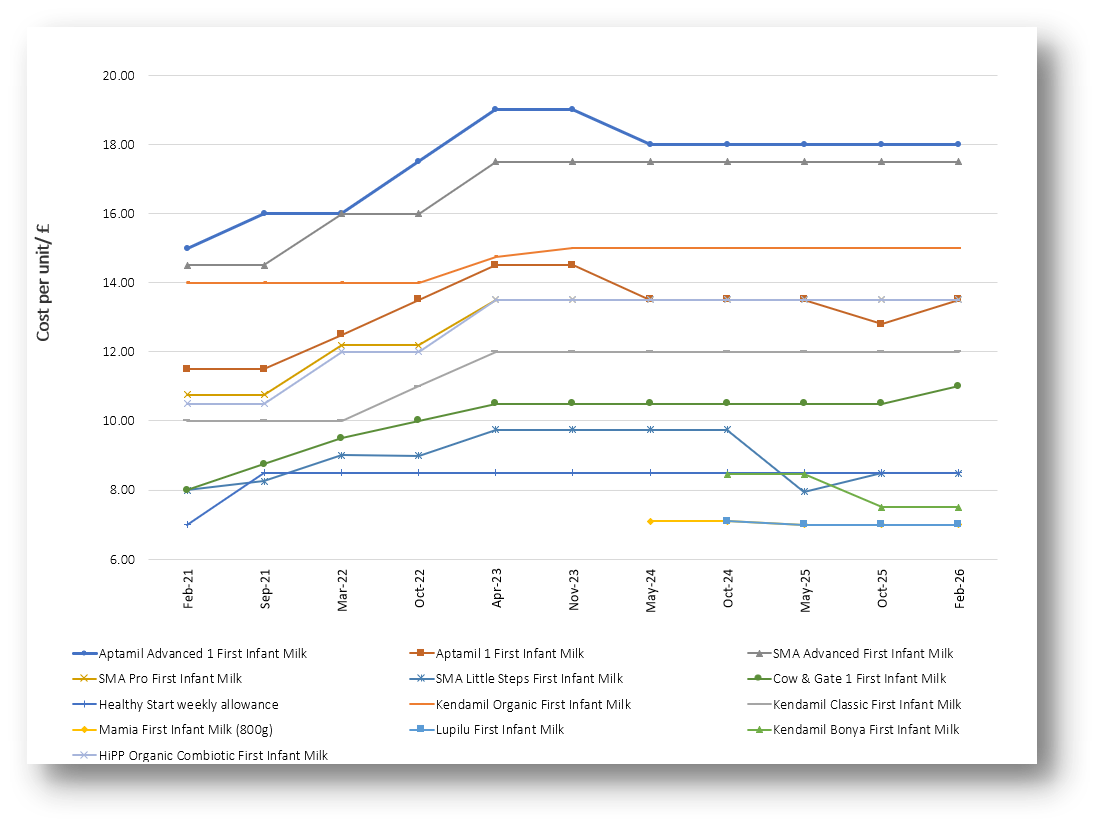
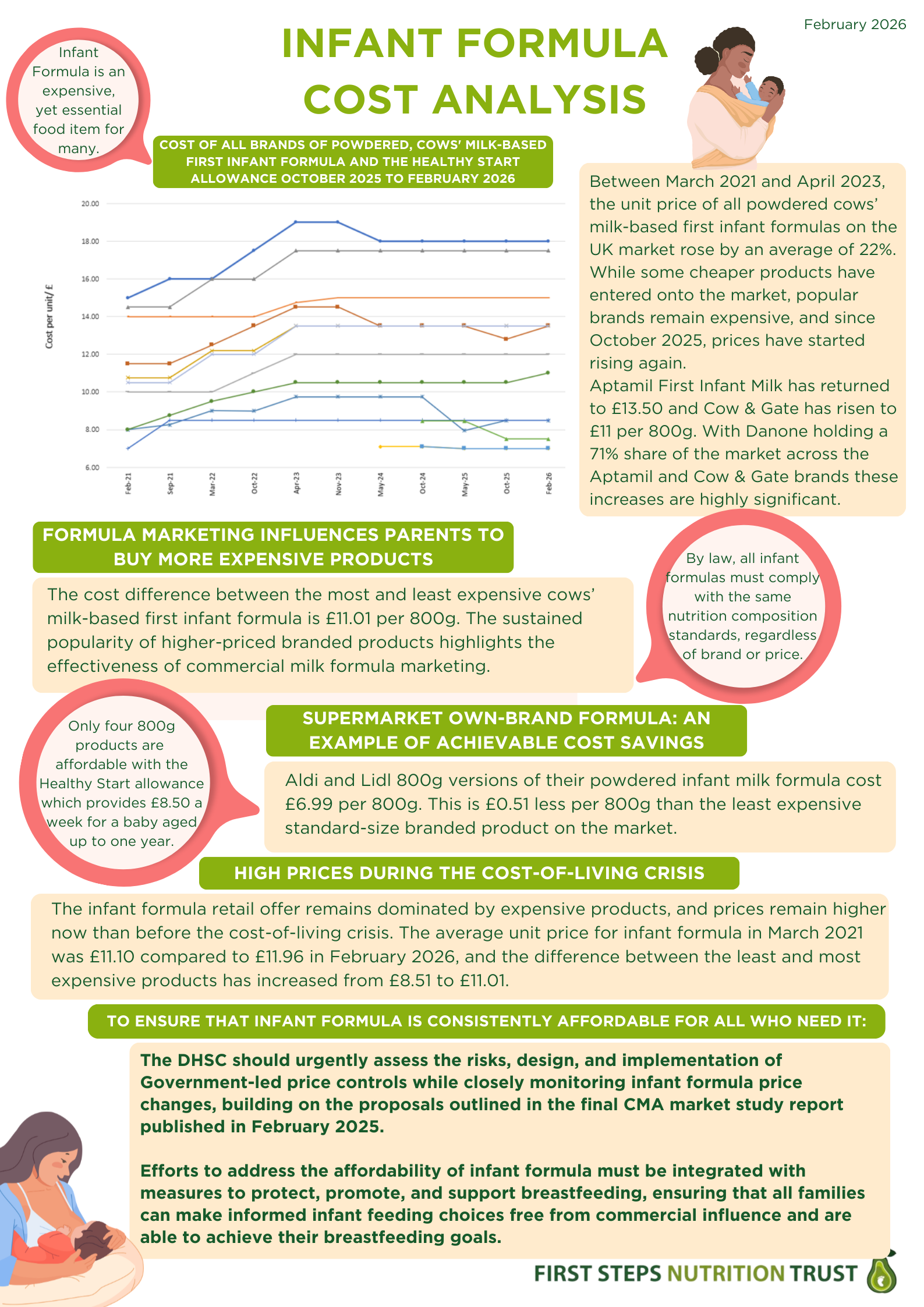
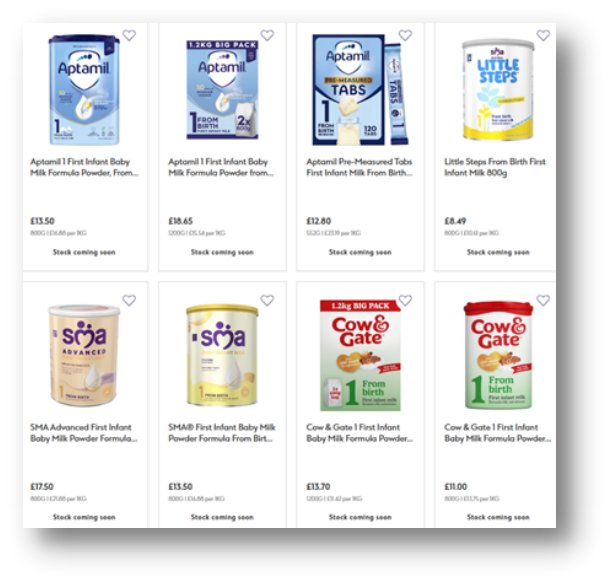
At the end of February, we published updated costs and trends reports as an additional update to our usual biannual releases in May and October. We did this in response to sharp price increases following the recent formula milk recalls (see below). Parents and carers quickly felt the impact, with one reporting on social media that Cow & Gate rose from £9.20 to £11 in a single week. Our findings were reported on by The Sun, The Grocer and the BMJ. Beyond pricing, the recent recalls also disrupted availability (see below).
Since October 2025, prices have increased across several major brands, particularly those owned by Danone (Aptamil and Cow & Gate) and Nestlé (SMA), which were affected by the recall (see Graph 1). The average cost of branded powdered first infant formula now stands at £12.95 per 800g tin (6p higher than in October) compared with £6.99 for supermarket own-brand products. Notably, Aptamil First Infant Milk has risen back to £13.50, undoing its earlier price reduction and Cow & Gate has increased to £11 per 800g, its highest recorded price since our monitoring began in March 2021. Although only a small number of products have risen in price, Danone and Nestlé together account for around 85% of the market, making these price increases significant.
Graph 1. Unit cost of powdered, cows' milk-based first infant formula and the Healthy Start allowance, March 2021 to February 2026
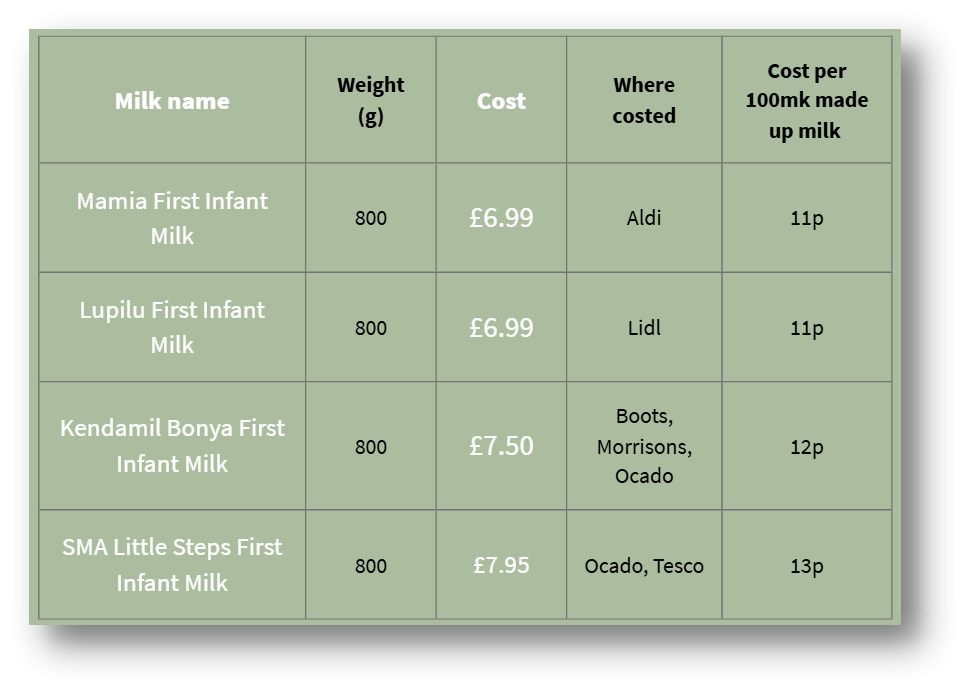
Only four of the 12 standard 800g infant formulas available cost less than the current £8.50 weekly Healthy Start allowance for babies under one, a figure unchanged since May 2025 (see Table 1). Two of these are supermarket own-brand products, which are typically less widely available. Although the allowance will rise to £9.30 in April 2026, this will still not increase the number of full-size tins families can afford at current prices.
Table 1. Lowest cost standard size (800g) infant formula on the UK market in February 2026
Furthermore, larger 2 x 600g packs of Aptamil, Cow & Gate and SMA Pro First Infant Formula, previously seen as more cost-effective purchasing options, have increased by between 50p and £2.65 since October 2025, reducing the value of these bulk-buy options. For example, exclusively formula feeding a 10-week-old baby with two large packs of Cow & Gate First Infant Milk now costs £41.40 per week, up from £36 in October.
Our analysis found that prices for products marketed as specialised milks are rising placing significant financial pressure on families; both those with a baby with a diagnosed clinical need and those influenced by misleading marketing in a weakly regulated market. It’s important to remember that some formulas marketed as specialised milks lack evidence of effectiveness (i.e. comfort milks and hungry milks) while others are recommended in clinical guidelines (i.e. anti-reflux and lactose-free) but should only be used under medical supervision. Specialised formula should only be used following formal diagnosis and on the advice of a healthcare professional, with appropriate clinical supervision and ongoing review.
Prices of ready to feed starter pack of infant formula prices fell between October 2025 and February 2026, likely reflecting the recent Government recommendation that healthcare settings provide infant formula that is competitively priced on the retail market. However, and importantly, these lower upfront prices do not necessarily translate into lower long-term costs. Manufacturers may price starter packs competitively to encourage brand loyalty, while charging more for the equivalent powdered formula used longer term.
Pricing strategies continue to mislead parents into perceiving expensive formulas as superior. The gap between the cheapest and most expensive 800g tin has widened to £11.01 (from £8.51 in March 2021), with prices now ranging from £6.99 to £18. Despite this variation, by law, all infant formulas must meet the same nutritional composition standards, regardless of brand or price and parents can be reassured that it is safe to switch between first infant formula brands.
Adding further complexity for families, retail prices for identical products remain inconsistent across supermarkets, with the retailers offering the lowest prices changing over time.
What needs to happen next
Our findings highlight the financial strain and uncertainty facing families who rely on formula to feed their babies. High and variable pricing, combined with misleading marketing strategies, risks undermining informed decision-making and may contribute to unsafe feeding practices.
In its final market study report published in February 2025, the Competition and Markets Authority proposed that price caps could act as a backstop measure to prevent excessive pricing, although it stopped short of recommending immediate implementation. We therefore strongly urge the Government to assess urgently the risks, design and implementation of potential price controls, so they are ready to act if prices remain high or continue to rise.
Action on formula affordability must sit alongside measures to protect, promote and support breastfeeding. All families should be able to make informed infant feeding decisions, free from commercial influence, and all women should be supported to meet their breastfeeding goals.
Further resources
Below are our two updated infographics, which can be found here on our website. ‘Infant formula cost analysis’ summarises the price trends and our asks of policy makers. ‘Lowest cost infant formula’ shows just that, and is intended to inform parents using formula to feed their babies if they are struggling with the cost.
Update: UK formula milk recalls
As highlighted in our February newsletter, the Food Standards Agency (FSA) issued alerts earlier this year regarding several formula milk products that were contaminated, or potentially contaminated, with the toxin cereulide, which can cause rapid-onset symptoms including nausea, vomiting, diarrhoea and abdominal cramps.
Health care practitioners should continue to advise parents who use formula to feed their babies to check for affected batches and avoid using any recalled products. Comprehensive recall lists, including pack sizes and best-before dates, as well as guidance for professionals, are available here.
Practitioners should also reinforce that powdered infant formula is not sterile. Parents and carers should be encouraged to carefully follow NHS guidance when preparing feeds. A key part of this guidance is using water that has been boiled and then allowed to cool to no less than 70°C before mixing with the powder, as this helps to kill any bacteria that may be present.
Our February formula cost analysis (reported above) identified a significant impact on formula milk availability following the recall. Several supermarkets temporarily withdrew affected Danone (Aptamil and Cow & Gate) and Nestlé (SMA) products, restricting families’ options. At the height of disruption, the Grocer, reported out-of-stocks of 28.4% (98 of 335 products).
Although supply has since improved, many families experienced difficulties. In response, we developed an infographic to support parents and carers, struggling to access their usual formula, available here.
Infant formula remains a necessary and appropriate option for some infants. It is therefore essential that the UK Government takes all necessary steps to ensure product safety and protect the health of vulnerable babies. We are convening with peers to assess this evolving and complex situation and have reached out to the Food Standards Agency to clarify the actions being taken to protect infant health.
For infant milk information please visit our website www.infantmilkinfo.org. If you can’t find what you’re looking for please email rachel@firststepsnutrition.org
Baby Feeding Law Group news
BFLG-UK statement on the Governments’ joint response to the CMA recommendations, and an update on DHSC progress and implementation
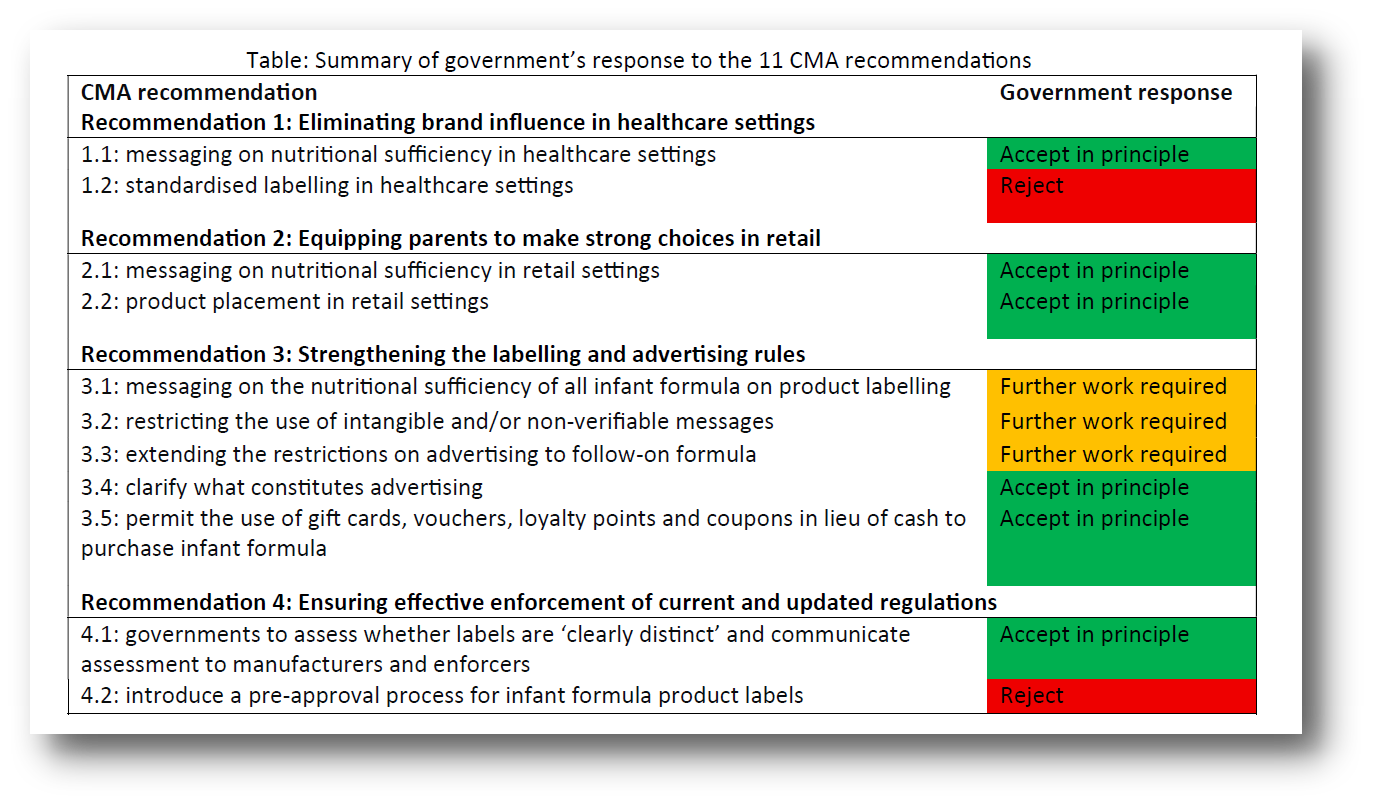
As shared previously in our February newsletter, on 3 December 2025, the Government published its joint response to the Competition and Markets Authority (CMA) infant and follow-on formula market study. On 19 February 2026, we published our BFLG-UK statement on this joint response.
We welcome the government’s acknowledgement of the significant, longstanding issues within the formula market in the UK related to marketing, pricing and retail practices which undermine public health recommendations on infant feeding. The government response makes several notable, positive commitments but further actions will be needed in the short term to deliver significant positive impacts for families and babies, especially those on low incomes. From the 11 recommendations made by the CMA, the government accepted six “in principle”, committed to “further work” on three, and rejected two:
The BFLG-UK statement outlines the steps we welcome and support; steps where greater ambition and urgency is required by the government and what we feel needs to happen next:
We welcome and support:
1. The government commitment to supporting babies, new parents and carers to eat well.
2. The government focus on supporting more informed choices by parent (recommendations 1.1, 2.1).
3. The government’s commitment to clarify what constitutes advertising (recommendation 3.4).
4. The government’s commitment to ensure labelling complies with the law (recommendation 4.1).
Where greater ambition and urgency by the government is required:
1. Strengthening formula labelling and advertising rules (recommendations 3.1, 3.2 and 3.3)
2. Ensuring effective enforcement (recommendation 4.2)
3. Eliminating brand influence in health care settings (recommendation 1.2)
4. Monitoring and intervening to bring down the cost of infant formula (backstop recommendation)
What needs to happen next:
Timely implementation of the 6 CMA recommendations which have been accepted in principle. We have requested more information on the specific actions and timeframes to achieve the above, as well as the indicators which will be used to chart progress and impact. This will then determine when and whether further steps are required.
Clarification on the next steps to strengthen labelling and advertising rules, with a clear timeline for the 3 recommendations (3.1, 3.2 and 3.3) for which further work is required and how this further work will be conducted. The goal should be stronger regulations in line with ‘the Code’, which places restrictions on the advertising of all formula milks marketed for use up to 3 years of age, and their proper enforcement. This would put children’s wellbeing above commercial interests.
Active monitoring and reporting on infant formula prices and commitment to undertaking a formal assessment of the need for and feasibility of implementing more direct measures to improve formula affordability such as profit caps (see above).
We have communicated with the government (including the DHSC and devolved nations) our willingness to support, to ensure that every family has access to affordable products and services they need to ensure the best start in life for their child – and that they are protected from insidious marketing practices that skew feeding choices.
We have met with a representative from the DHSC, and they have shared that they are prioritising delivery of actions, especially those committed to in the government’s manifesto. The DHSC also shared how they are addressing the steps required to implement the government response in their perceived order of priority, including how they are working with and delegating responsibilities to the devolved nations (Food Standards Agency Northern Ireland, Food Standards Scotland and Public Health Wales) and other stakeholders involved in the actions required (for example, the NHS supply chain).
Considering our most recent infant formula costs and trends analyses which show increases (see above), we remain concerned that current actions are insufficient to ensure that infant formula is consistently affordable for parents and families. We therefore continue to advocate for the DHSC to urgently assess the risks, design and implementation of Government-led price controls while closely monitoring infant formula price changes, building on the proposals outlines in the final CMA market study report of February 2025.
For more information about the BFLG-UK please visit our website Baby Feeding Law Group UK (bflg-uk.org) and sign up to our twitter (X) account @BflgUk. You can also email katie@firststepsnutrition.org
Forthcoming
First Steps Nutrition Trust 2025 impact report
We’ve been busy writing our 2025 impact report and have created this infographic below which provides a snapshot of our top achievements of last year. The full report will be available next month. A huge thank you to our generous donors without which our work would not be possible. We are working hard to secure sustainable and more diverse income sources to secure the future of First Steps Nutrition, so please be in touch if you think you can help.
Imperial Ultra-Processed Food Policy Forum, hybrid, March 24th
Following up from 2024’s popular event, the team at Imperial College are hosting another one day UPF food policy forum, assessing the science and advancing regulatory and policy responses to ultra-processed foods. The event convenes world-leading scientists, politicians, policymakers and civil society representatives (including our Director, Vicky) at Imperial College London. Together, they will discuss the latest scientific evidence on UPFs and explore the scope for effective, evidence-based regulatory and policy responses to their growing dominance in people’s diets and their impacts on health.
This year’s Forum will focus on accelerating progress towards a more robust, science-based policy agenda. As governments worldwide intensify efforts to address the expanding role of UPFs in diets, high-quality research is increasingly central to shaping policies that are both effective and sustainable.
Momentum for action in the UK and internationally has continued since the 2024 Imperial event. A series of papers published in The Lancet in November 2025 marked a turning point, providing a strong framework for bridging science and policy (read more in our December newsletter).
The Ultra-processed Food Policy Forum will showcase policy approaches from across the globe, providing policymakers, researchers and civil society with comparative insights and practical lessons for future action. Vicky’s presentation will focus on early years nutrition.
Find out more and register to attend HERE.
An update of First Steps’ Eating Well Healthy Start and Best Start Foods, a practical guide
Next month will see a small but welcome 10% increase in the monetary allowance provided to Healthy Start beneficiaries. To ensure our Eating Well resource Healthy Start and Best Start Foods – A practical guide remains current we will be updating this resource to reflect this increase and other changes in public health recommendations and the wider context since 2022. We will also be sharing new ‘bite sized’ resources, likely including posters, leaflets and recipe cards for use in Family Hubs and food bank settings. Watch out for these new resources in our future newsletters.
The iHV Evidence Based Practice conference, Bournemouth, May 6th
The Institute of Health Visiting (iHV) will be hosting a full-day Evidence-based Practice Conference 2026, with the theme “From Evidence to Action: Getting it right from the start”, at Bournemouth International Centre on Wednesday 6 May 2026. Our Director, Vicky will be doing an oral presentation on Starting solids and the latest public health recommendations on shop bought baby foods. You can book your tickets here before this Saturday, March 7th.
HR News
Welcoming Sara
We’re excited to welcome recently qualified dietitian and MSc Nutrition graduate Sara Esser on to our team as our new Nutritionist. Sara brings with her several years of experience working in public health nutrition with the UCL Healthy Weight Policy Research team. Through her dietetic placements in NHS paediatric settings and her voluntary work supporting vulnerable families, she saw first‑hand the importance of addressing health inequalities to improve early years nutrition and long‑term health, which drew her to working for First Steps. She has particular interests in infant and young child nutrition, the regulation and marketing of breastmilk substitutes, and translating complex guidance into clear, practical information for families and practitioners.